Eomyodon volkeri
Eomyidae is a scarcely known family of extinct rodents. The natural history of the family is practically based solely on dental, mandibular and limited cranial remains. However, Eomyodon volkeri is one of the very rare specimens with postcranial material preserved.

The authors that described the specimen (Engler & Martin 2015) state that, based on several limb indices, and no gliding membrane present (as opposed to the closely related, gliding Eomys), it was a terrestrial, ground-dwelling form with a rather generalized proportions for a rodent. The estimated weight is around 10 g. While the preserved specimen indicates considerable fur coverage, the individual hairs and stomach content remains unidentifiable. It lived in the swamp and mesophytic forests in the vicinity of a large, isolated maar lake with an anoxic bottom, which with numerous other fauna enabled the exceptional state of preservation. It lived during the Late Oligocene (MP28, ~24Ma), in present day Germany.
References:
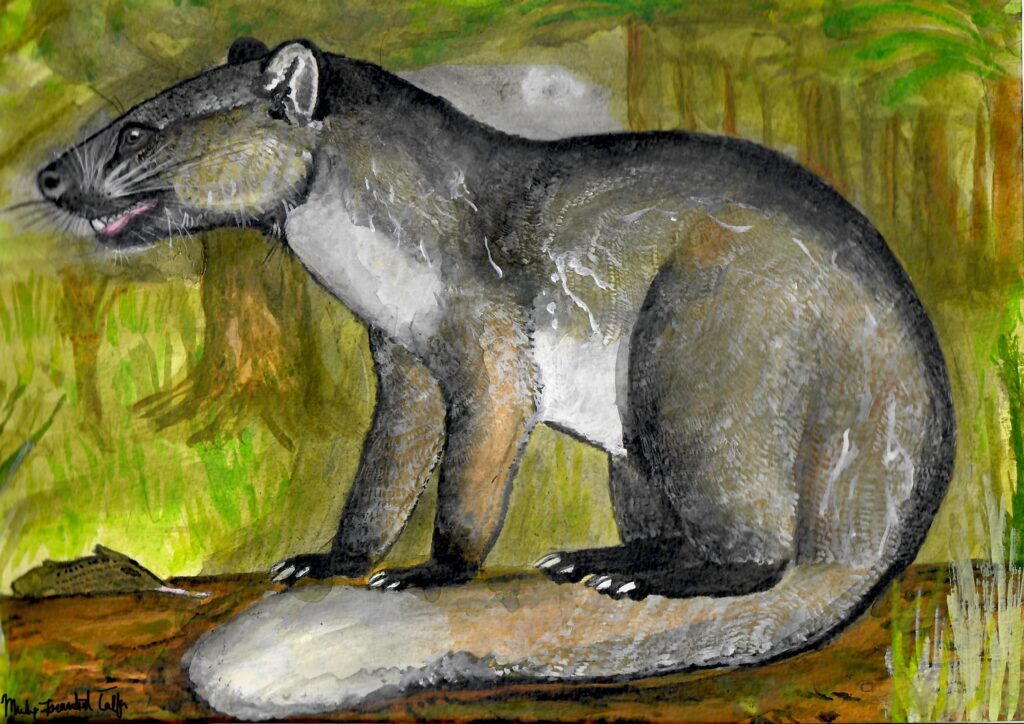
Masrasector nananubis
My life reconstruction of Masrasector nananubis, an Egyptian, Late Eocene (Priabonian, ~34 Ma) teratodontine hyaenodont of the size of a mongoose (roughly 1 kg). As described by (Borths & Seiffert 2017), based on the morphology of the distal humerus, it was an agile, terrestrial predator.
References:
Apterodon langebadreae
Here I present my life reconstruction of Apterodon langebadreae, a semiaquatic hyainailourid hyaenodont of Middle Eocene Libya. As described by (Grohé et al. 2012), based on the limb morphology, this species possessed pronounced adaptations for digging as well as swimming. Moreover, like all the other members of Hyainailouridae, Apterodon had a proportionally massive head and different body proportions compared to modern carnivorans. In the reconstruction, it is depicted at the edge of a riverbank, accompanied by a few crustaceans as potential prey.

References:
List of fossil material attributed to scaly-tailed squirrels (Anomaluroidea, Rodentia)
This article is a synthesis of referenced literature on the topic, and apart from my life reconstructions I have not conducted any original research on the group. I wrote it in a way that it may resemble an actual scientific article a little, so it helps to highlight and organize information in a better manner. If you are aware of any paper I have not referenced, please contact me, so I can make this list even more comprehensive.
Anomaluroidea is a poorly studied superfamily of elusive rodents of mainland African origin and present zoogeographical distribution. This study presents a synthesis of all dental and skeletal remains attributed to Anomaluroidea throughout the tertiary, with an introduction to the ecomorphology, evolution and systematics or the group. In total 216 specimens in 5 families are included in the list.
Introduction to the group’s taxonomy, phylogeny and morphology:
The superfamily Anomaluroidea contains in total of 7 extant species distributed in 2 families. Anomaluridae includes the subfamilies Anomalurinae and Idiurinae. Anomalurinae includes 4 species of scaly-tailed squirrels, the Lord Derby’s scaly-tailed squirrel (Anomalurus derbianus), Pel’s scaly-tailed squirrel (Anomalurus pelii), dwarf scaly-tailed squirrel (Anomalurus pusillus) and Beecroft’s flying squirrel (Anomalurus beecrofti) which is the most divergent of the anomalurines and is sometimes included in its own genus Anumalurops (Fabre et al. 2018). Idiurinae includes the long-eared flying mouse (Idiurus macrotis) and the pygmy scaly-tailed flying squirrel (Idiurus zenkeri) (Schunke et Hutterer 2017). The family Zenkerellidae includes only a single species, the Cameroon scaly-tail (Zenkerella insignis), which has not been directly observed by a western mammalogist until 2017 (Dinets, 2017).Together with the springhares (Pedetidae), they form the suborder Anomaluromorpha, one of the four main suborders of rodents (Fabre et al. 2018). Based on palaeontological and molecular evidence, Anomaluroidea is a remarkably ancient and conservative clade, with the divergence time of the crown group Anomaluroidea being estimated during the early or middle Eocene (Heritage et al. 2016). Originally, the Zegdoumyidae, recorded from the Middle Eocene, was thought to be the basalmost anomaluroid family (Marivaux et al. 2014). There is some uncertainty regarding their anomaluroid affinities, however (Coster et al. 2015). Later, using the Bayesian tip-dating analysis, the springhare (Pedetidae), an anomaluromorph but not an anomaluroid (scaly-tailed squirrel) was found to be more closely related to Anomaluroidea than the Zegdoumyidae, therefore placing Zegdoumyidae as stem Anomaluromorpha (Seiffert et al. 2024). This then excludes them from the list.


Extant anomalurids posses several unorthodox gliding adaptations not recorded in any other living volant mammal (colugo, pteromyin squirrels and sugar gliders) Among these, a specialized rod-like cartilage on the arm, originating from the olecranon proccess of the proximal ulna (Amador, 2022), and peculiar scales on the ventral side of the proximal part of the tail. (Panyutina et al. 2020). Furthermore, anomaluromorphs (Anomaluroidea and Pedetidae) possess the orbital portion of the infraorbital part of the zygomaticomandibularis as well as the rostral portion, hence exhibiting double origin of the zygomaticomandibularis. The relevance of this trait is in the fact that the orbital portion of the zygomaticomandibularis was thought to be absent in rodents (Druzinsky et al. 2011), and while it has been eventually confirmed in Bathyergidae (Cox & Faulkes, 2014; Cox et al., 2020), the later is not of double origin as in the former. Therefore, this structure of the infraorbital part of the zygomaticomandibularis is unique to Anomaluromorpha (Da Cunha et al. 2021).
Materials and methods:
The data in the list were gathered from published literature spanning the period 1973-2024. Included were papers, books, dissertations, abstracts, and posters, for a total of 24 publications. They were found using Google Scholar or searching for author’s publications online. Any possible unpublished, privately-owned or undescribed specimens were excluded. Individual specimens and localities are not cited separately if the species was published in a single paper. In such case, the authors are cited solely at the begging of the list of a given species. The life reconstructions were done traditionally, using paper and a mechanical pencil, and then colored digitally. With the smaller species (Shazurus, Argouburus), allometric growth was considered, which directly affects the body proportions – head to body size ratio. The cranial and postcranial proportions of each species were reconstructed based on its closest living relative, i.e. Zenkerella, Anomalurus, or Idiurus. Original specimens were digitally scaled to the nearest 0.1 mm.
Results:
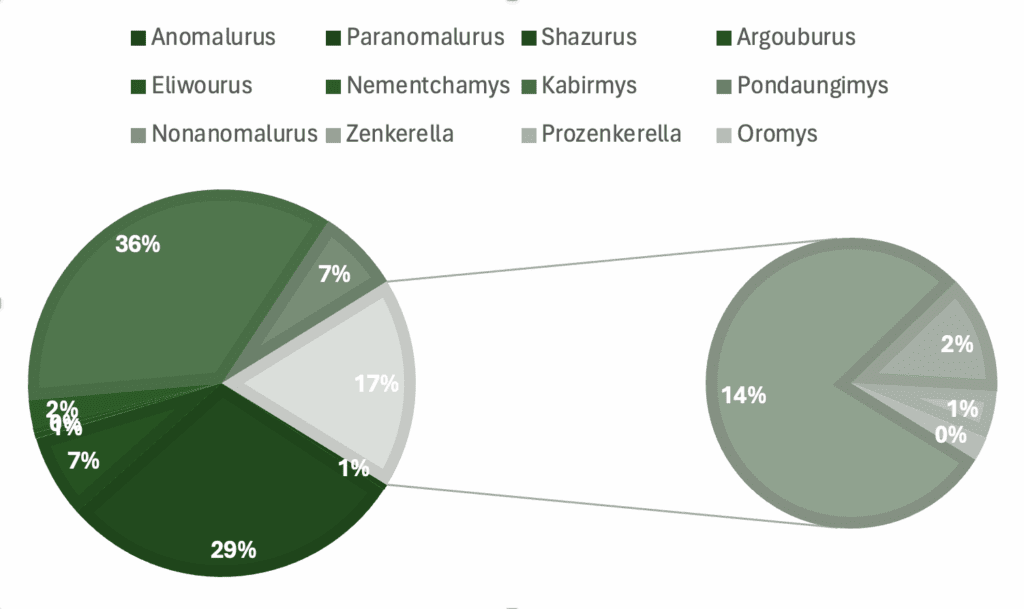
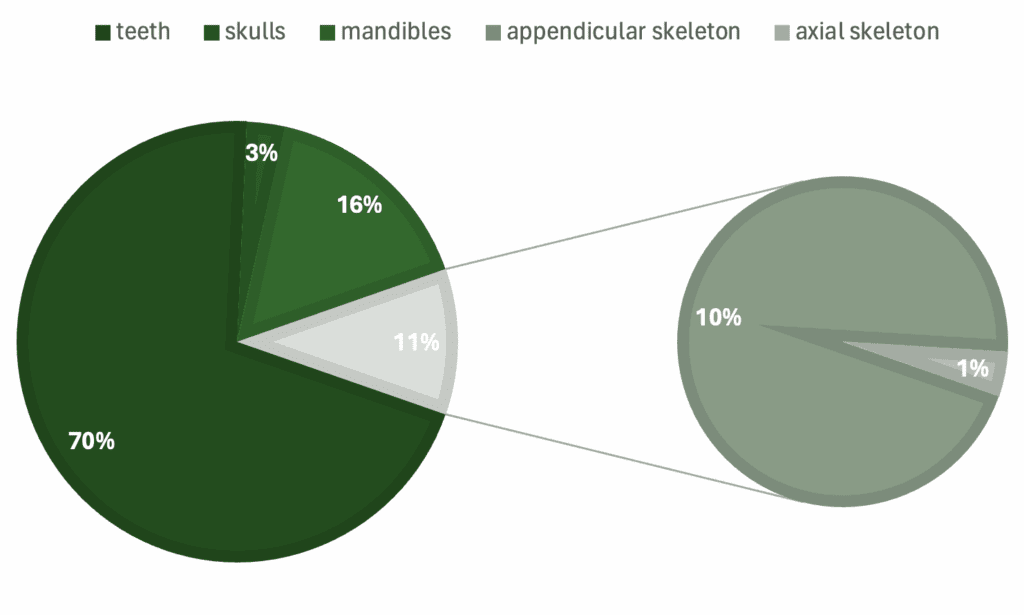
Graphs showing percentage of specimens regarding a) genera, b) skeletal material, c) country of origin, d) epoch.
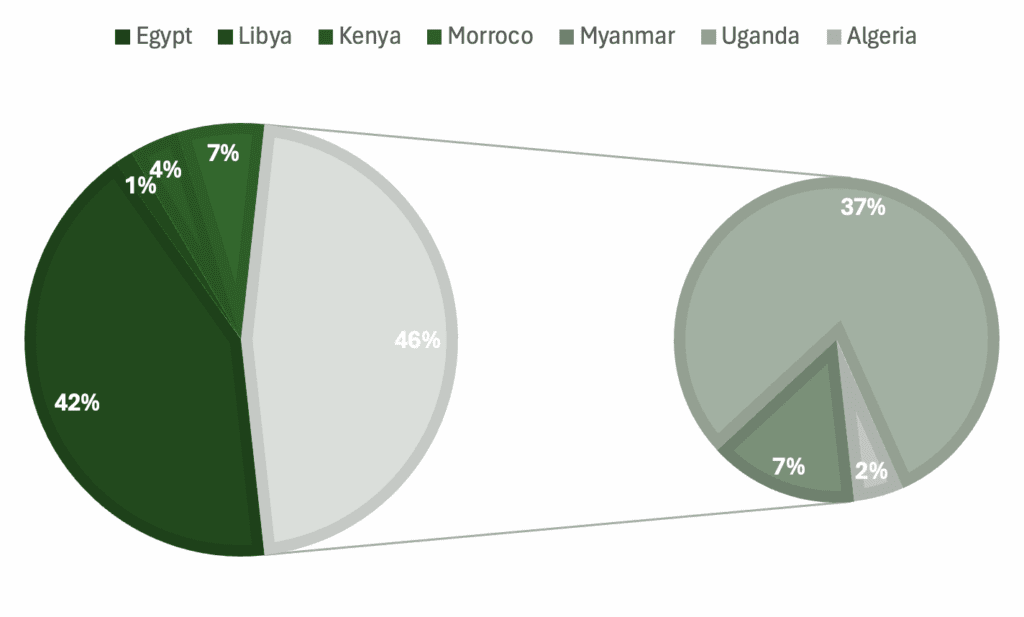
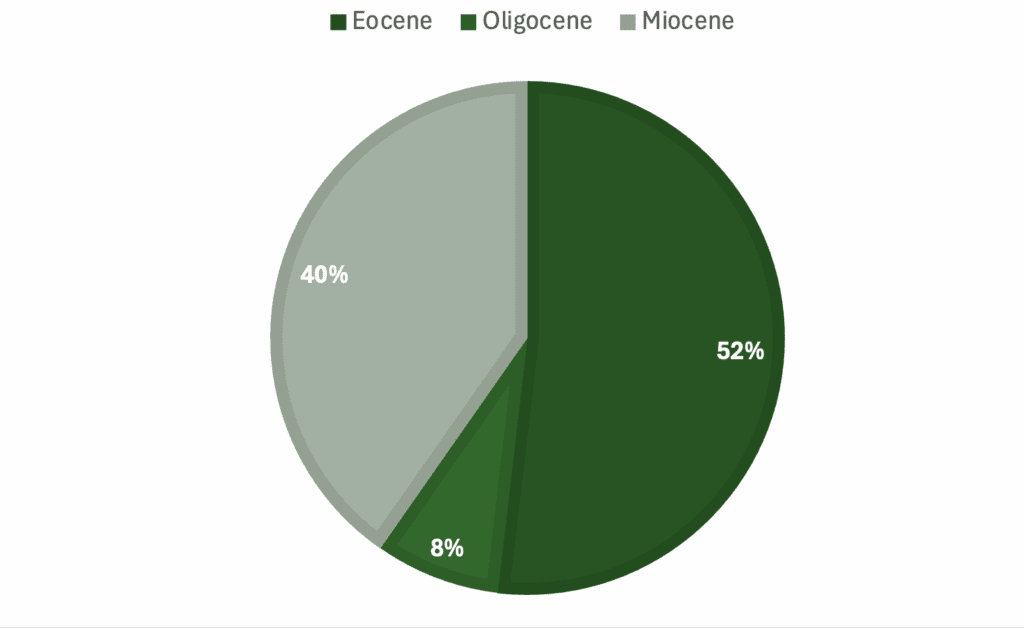
List:
Anomaluridae
Family that includes all the extant gliding anomaluroids, Anomalurus and Idiurus. Members of this group possess a rod-like structure originating from the proximal ulna that enchances gliding locomotion, it is a unique structure only confined to this clade. Some of the species included in this family in the list are not explicitly described in cited literature as belonging to Anomaluridae sensu strictio, but are very frequently compared to the latter based on the profound dental similarities. That is why they are listed under this family.
Anomalurus (Waterhouse, 1843) – middle Miocene, Kipsaraman, Muruyur Beds, Kenya; in total, 1 fossil specimen listed.
(Winkler et al. 2010)
KNM-TH 19439 – left M3,
Paranomalurus bishopi (Lavocat, 1973) –Early Miocene (approximately 20 Ma); Napak IV, V, XV, XXX, XXXI, Northeastern Uganda; the largest species of Paranomalurus, volant, generally thought of as phenotypically very similar to the extant Anomalurus; in total, 43 specimens listed.
(unable to provide description of the type specimen, see (Lavocat, 1973))
(Winkler et al. 2010)
BUMP 82 – neurocranium, right p4-m3
(Bento da Costa; 2020) – no description to individual specimens provided
Teeth – NAP IV 76’09; NAP IV 5’12; NAP IV 165’12; NAP V 9’04; NAP V 191’08; NAP V 246’11; NAP V 146’13; NAP V 7’15; NAP V 49’16; NAP V 83’16; NAP XV 97’09; NAP XV 142’09; NAP XV 155’09; NAP XV 57’14; NAP XV 25’16; NAP XXX 196’14
Mandibles – NAP I 308; NAP I 42’99; NAP IV 50’06; NAP IV 58’08; NAP IV 78’10; NAP IV185’12; NAP IVlower 210’12; NAP IV 25’18; NAP IV 95’18; NAP V 65S; NAP V 24’02; NAP V 36’06; NAP V 25’10; NAP V 98’15; NAP IX AUG 62; NAP XV 171’07; NAP XV 172’07; NAP XV 165’08; NAP XV 53’09; NAP XV 43’10; NAP XV 33’14; NAP XV 16’15; NAP XV 73’16
Ulnae – NAP IV 185’11; NAP V 51’03; NAP XV 64’12
NAP XV 113’07 – right distal humerus

Paranomalurus walkeri (Lavocat, 1973) – Early Miocene (approximately 20 Ma); Napak IV, V, XV, XV, XV, XXXI, Northeastern Uganda; dentition similar to P. bishopi but smaller in size, volant, generally thought of as phenotypically very similar to the extant Anomalurus; in total 13 specimens listed.
(unable to provide description of the type specimen, see (Lavocat, 1973))
(Bento da Costa; 2020) – no individual description to specimens provided
Teeth – NAP IV 64 428; NAP IV 108’13; NAP IV 68’14; NAP V 145’13; NAP V 172’15; NAP XV 146’09; NAP XV 6’12; NAP XV 36’18; NAP XXXI 56’16
Mandibles – NAP XV 146’09; NAP XV 6’12
Femora – NAP XV 375’08; NAP XV 99’09
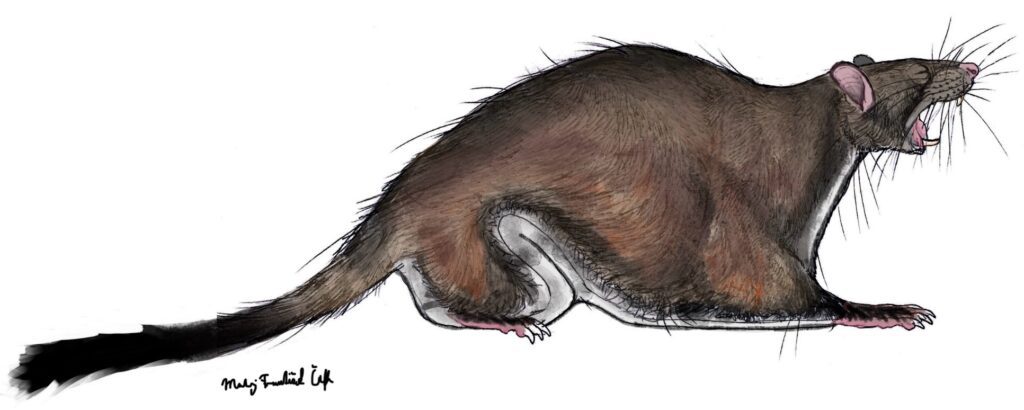
Shazurus minutus (Sallam et al. 2010) – Earliest Late Eocene (approximately 37 Ma); Birket Qarun Locality 2 in the Fayum Depression of northern Egypt; a miniscule species of an anomalurine rodent, with a dentition similar to Paranomalurus; in total, 15 specimens listed.
DPC 23305A – left p4; DPC 21294A – right p4; DPC 21747A – right p4; DPC 21488B – left m1; DPC 21220B – right m1; DPC 21293L – left m1; DPC 21296A – right m2; DPC 21500B – right m2; DPC 21358A – left m2; DPC 22442B – left m2; DPC 23305B – right P4; CGM 83701 – left M1; DPC 21538A – right M1; DPC 21221A – right M2; DPC 21452A – left M2
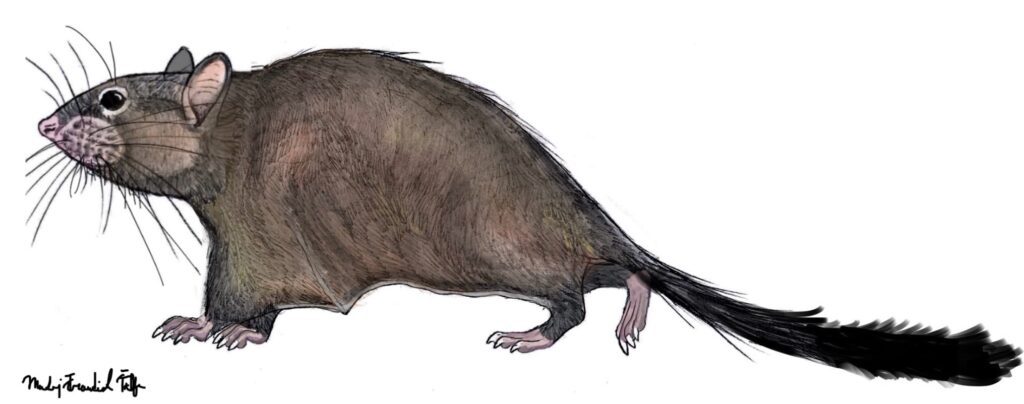
Argouburus minutus (Marivaux et al. 2017) – Earliest Oligocene (approximately 33 Ma); Upper Samlat Formation, westernmost part of the Sahara, Morroco; a miniscule species, differs from both Shazurus and Paranomalurus in lacking the Y-shaped structure of the third and fourth transverse crests; in total, 1 specimen listed.
DAK-Arg-010 – Left M3; holotype

Zenkerellidae
Non-volant, scansorial family, including the sole extant species, the Cameroon scaly-tail (Zenkerella insignis). The behavioral ecology of this species is poorly understood (Heritage et al. 2016).
Zenkerella wintoni (Lavocat, 1973) – late Early Miocene (approximately 17 Ma); Moroto II, Kogole, Songhor, Kenya; smaller than Z. insignis; In total, 5 specimen listed.
mandible – (unable to provide more detailed description of the specimen, see (Lavocat, 1973))
(Pickfort et. Mein 2006)
Mor II, 14’98 – left mandible fragment with m1 and m2; Mor II, 33’03 – right molar fragment (left m3?); Mor II, 46’04 – left P4; Mor II, 47’04 – left p4

Prozenkerella saharaensis (Coster et al. 2015) – Early Oligocene; Zallah 7 Incision locality, Continental and Transitional Marine Deposits (CTMD), central Libya; a species very similar to Zenkerella wintoni and Zenkerella insignis, larger than Z. wintoni but slightly smaller than Z. insignis; in total, 2 specimens listed.
Z7I-12 – right m, holotype; Z7I-13 – right M

Oromys zenkerellinopsis (Marivaux et al. 2017) – Earliest Oligocene (approximately 33 Ma); Upper Samlat Formation, westernmost part of the Sahara, Morroco; miniscule species with brachydont and quadrangular teeth, differs from other Zenkerillidae in showing a less-rounded crown outline; in total, 1 specimen listed.
DAK-Arg-009 – left M1 or M2, holotype

Nonanomaluridae
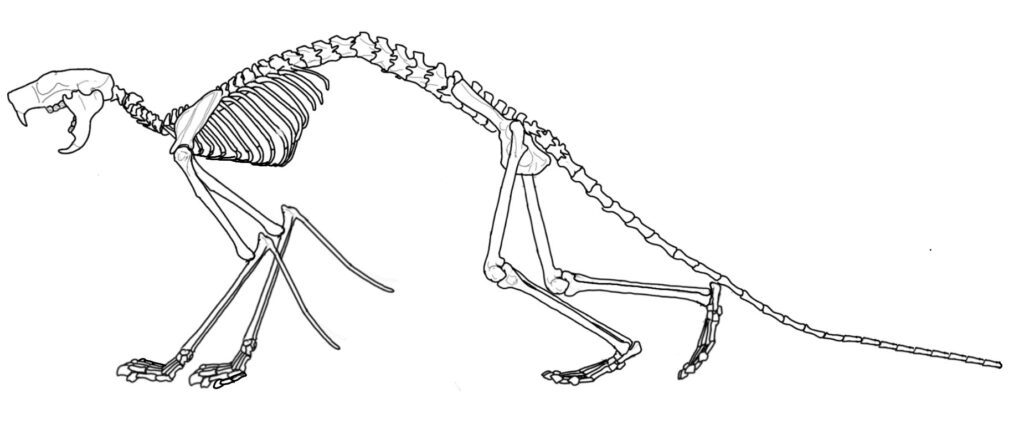
A monotypic family including only the genus Nonanomalurus. The family exhibits postcranial anatomy lacking the typical distal elongation of the appendicular skeleton of the gliding forms such as Anomalurus, being the result of an arboreally and partially terrestrially adapted mode of life without gliding (Pickford et al. 2013).
Nonanomalurus soniae (Pickford et al. 2013) – Uganda Early Miocene (approximately 20.5-18.5 Ma); Napak IV, V, XV, XXX, XXXI, Akisim remnant of the Napak Volcano, Northeastern Uganda; non-volant arboreally adapted species retaining several plesiomorphic characters shared with Pedetidae. Postcranial proportions drastically different from contemporaneous anomalurids, possibly indicating a lifestyle similar to extant galagid primates; in total, 25 specimens listed.
(Lavocat, 1973)
KNM SO 788 – left maxilla P4-M2; holotype
(Pickford et al. 2013)
Nap XV 170’07 – partial skeleton: skull, mandibles, the vertebral column, rib cage, both scapulae, complete left humerus and radius and parts of the ulna, broken right humerus, ulna and radius and disarticulated wrist and manual bones (which are close together) a damaged pelvis, complete left femur and tibia, broken right femur and tibia, talus, ankle and pedal bones. Inside the thoracic cage, four partially ossified long bone diaphyses (two femur and two tibiae) an isolated frontal bone, and an incompletely formed otic bulla, which the authors interpret as the remains of a fœtus (less likely a new born infant).
(Bento da Costa; 2020) – no individual description to specimens provided
teeth – NAP I 58 308; NAP IV 52’07; NAP IV 60’09; NAP IV 14’1; NAP V 8’04; NAP V 140’11; NAP V 245’11; NAP V 83’12; NAP V 155’14; NAP XV 36’11;
postcranial material – NAP I 135’99; NAP V 20’98; NAP V 6’01; NAP V 49’10; NAP V 83’12; NAP XIII 8’14; NAP XV 201’08; NAP XV 59’16; NAP XXX 23’15; NAP XXXI 50’14; NAP XXXI 58’18; NAP XXXII 6’16; NAP XXXII 25’16

Nonanomalurus parvus (Marivaux et al. 2017) – Earliest Oligocene (approximately 33 Ma); Upper Samlat Formation, westernmost part of the Sahara (Morocco); half the dental size of N. soniae, differs from itin the morphology of the upper molars for instance having lower, shorter and thinner transverse crests; in total, 5 specimens listed.
DAK-Arg-011 – left M1, holotype; DAK-Arg-007 – right M2; DAK-Pto-005 – right P4, lingually broken; DAK-Arg-003 – right m2; DAK-Arg-004 – left p4
Nementchamyidae
The basalmost family included in Anomaluroidea. The family exhibits a generalized postcranial morphology, indicating an adaptation to a non-volant lifestyle (Coster et al. 2015; Coster et al. 2019).
Kabirmys qarunensis (Sallam et al. 2010) – Earliest Late Eocene (approximately 37 Ma); Birket Qarun Formation, Umm Rigl Member, Egypt; the largest known anomaluroid, lacking extensive enamel crenulation on the upper and lower teeth, non-volant; in total, 76 specimens listed.
CGM 83698 – right mandible with m1–2, holotype; CGM 08-289A – fragment of left mandible with dp4 and incisor; CGM 21839G – left mandibular fragment with m1; DPC 24494 – fragment of left maxilla with P4 and M1 alveoli; DPC 23305I – right mandible with m1 and alveoli for p4, m2, and m3; DPC 21839F – left M1 or M2; DPC 23305F – left M1 or M2; DPC 21502C – left M1 or M2; DPC 21538F – left M1 or M2; DPC 21221E – left M1 or M2; DPC 21296H – left M1 or M2; DPC 21452F – left M1 or M2; DPC 21500M – left M1 or M2; DPC 21293O – right M3; DPC 21221F – right M3; CGM 08-328B – left M3; DPC 21358G – left M3; DPC 21488G – left M3; DPC 23305F – left M1 or M2; DPC 21374A – left dp4; DPC 21452B – left dp4; DPC 21221B – left dp4; DPC 21360A – left dp4; DPC 21839A – left dp4; DPC 23305C – left dp4; DPC 21294B – right dp4; DPC 21538B – right dp4; DPC 21293M – right dp4; DPC 21358B- right dp4; DPC 21296E – left dp4 or p4; DPC 21500K – right p4; DPC 22442H – left p4; DPC 21839B – left p4; DPC 23305D – right p4; DPC 21538C – left m1; DPC 21221C – left m1; DPC 21294C – left m1; DPC 21358C – right m1; DPC 21360B – right m1; DPC 21374B – right m2; DPC 21452C – right m2; DPC 21500L – right m2; DPC 21538D – right m2; DPC 21839C – right m2; DPC 22442I – right m2; DPC 21360C – right m2; DPC 21221D – right m2; DPC 21296G – left m2; DPC 21294D – left m2; DPC 21306D – left m1 or m2; DPC 21374C – right m3; DPC 23305E – right m3; DPC 21452D – left m3; DPC 21358D – left m3; DPC 21839E – left dP4; DPC 21538E – left dP4; CGM 08-328A – right dP4; DPC 21220D – right dP4; DPC 21360E – right dP4; DPC 21294E – right P4; DPC 21371I – left P4; DPC 21488D – left P4; DPC 21452E – right M1 or M2; DPC 21347D – right M1 or M2; DPC 21747I – right M1 or M2; DPC 22442J – right M1 or M2; DPC 21220E – right M1 or M2; DPC 21358E – right M1 or M2; DPC 21458E – right M1 or M2; DPC 21374E – left M1 or M2; DPC 21371J – left M1 or M2; DPC 21293N – left M1 or M2; DPC 21294F – left M1 or M2; DPC 21358H – left M1 or M2; DPC 21360F – left M1 or M2; DPC 21488F – left M1 or M2

Kabirmys prius (Coster et al. 2015) – Late Middle Eocene (approximately 37 Ma); DT-Loc. 3, New Idam Unit of the Dur At-Talah escarpment, south- central Libya; approximately the same size as K. qarunensis but more plesiomorphic, species with brachyodont and pentalophodont upper molar; in total, 1 specimen listed.
DT3-1 – right M2; holotype
Nementchamys lavocati (Jaeger et al. 1985) – Late Eocene; Nementcha locality, southern slope of Nementcha mountains, Eastern Algeria; species of large size, with dentition typical of the family, strong anterolophid on lower molars, molarized P4, occlusal surface complex, with numerous enamel crenulations; in total, 5 specimens listed.
UON84-25 – right m1; holotype; UON84-26 – left p4/dp4; UON84-27 – right m3; UON84-28 – right P4; UON84-29 – left M3
Pondaungimys anomaluropsis (Dawson et al. 2003) – Late Middle Eocene (approximately 40Ma); Paukkaung locus no. 2 (PK2) and Saba Pondaung (PK4 – Spg), Pondaung area, Central Myanmar (Coster et al. 2019), Bhl locality, Bahin area, Central Myanmar (Dawson et al. 2003); the solely recorded anomaluroid from Asia, the dentition similar to other nementchamyids, non-volant; in total, 15 specimens listed.
(Dawson et al. 2003)
NMMP-KU 0213 – left mandibular fragment m1-3; holotype; NMMP-KU 0048 – right maxillary fragment with P3–4; NMMP-KU 0047 – left m2; NMMP-KU 0049 – left mandibular fragment with m2–3
(Marivaux et al. 2005)
Pkg 157 – partial skull with well-preserved left and right maxillae with P4–M3; Pkg 192 – right DP4; Pkg 193 – left P4; Pkg 194 – right m3; Pkg 195 – left p4; Pkg 196 – left m1; Pkg 197 – left m3; lingual and posterior borders broken; Spg 14 – right p4; Spg 14 – right m1; Spg 14 – right mandibular fragment with p4 and m1
(Coster et al. 2019)
MFP 2014-1 – left astragalus
Discussion and conclusion
The evolutionary history of Anomaluroidea reflects a remarkably ancient and conservative clade (Heritage et al. 2016), likely to be the oldest rodent clade from Africa, that has (to our current knowledge) diversified morphologically very little. Anomaluroidea might potentially represent a rare case of African autochtonous rodents dispersing to Asia as early as Middle Eocene (Coster et al. 2019), which is opposed to most rodent clades that dispersed vice versa, that is, from Asia to Africa, such as phiomorph rodents, which appear to be much more common in fossil localities with anomaluroids present (Marivaux, 2017). This hypothesis is in need of better clarification since there is still the possibility that anomaluroids originated in Asia and dispersed to Africa, with the genus Pondaugimys of Middle Eocene Myanmar representing the oldest undoubted occurrence of this clade, and the only described anomaluroid of Asia. If an asiatic origin of the Anomaluroidea is indeed correct, Zegdoumyidae might not be related to Anomaluroidea at all (Coster et al. 2015). The state of origin for Anomaluroidea shall remain open till more exhaustive material is known to science for better clarification.
The members of the family Nementchamyidae appear to be considerably large in dimension for anomaluroid standards (Jaeger et al. 1985; Sallam et al. 2010), and most likely did not have any adaptation for gliding (Sallam et al. 2010). The same applies for the monotypic Nonanomaluridae, of which the entire skeleton has been preserved, shedding light on its ecomorphology. Nonanomalurus was most likely predominantly arboreal, non-glissant form with partially limited terrestrial lifestyle as well (Pickford et al. 2013; Bento da Costa, 2020). Overall, anomaluroids are known to Eocene sites of North Africa while completely lacking in any Oligocene sites of the same region. The sympatrically known phiomorph rodents do not show this pattern of rehearsal, likely suggesting that a profound alteration of habitats was present in the region somewhere at the Eocene – Oligocene boundary (Sallam et al. 2010)
In conclusion, Anomaluroidea remains as one of the least known clades of extant mammals in the world, and the same applies for the fossil record and evolutionary history. Current picture of the natural history of anomaluroids is partial and incomplete. The aim of this study is to synthesize current record of the clade to acquire a more accurate undestanding of the present knowledge of this group. Until more fossil material is unearthed and empirical studies are done on the topic, not much more can be said about the ecomorphology, systematics, and behaviour of anomaluroids.
References:
Pika of Early Pleistocene Slovakia
Ochotona horaceki is a small species of pika of the subgenus Ochotona (theoretically could be considered as), related to the extinct steppe-adapted pikas that during the pleistocene inhabited most of Europe. These European forms are thought to be of the “O. pusilla” species clade, hence, their closest extant relative is unsuprisingly the steppe pika (Ochotona pusilla).
It was found in the western part of the Slovak Karst mountain range, and described in 2004 as a new species based on several unique traits. It lived during the Early Pleistocene (Calabrian), Biharian Small Mammal age.

References:
Rhombomylus and Matutinia, basal gliroids – a poorly understood group of early mammals
It is time to take a dive into more very scarcely understood “rodents” of Eocene China, to actually see how they might have looked like when they were still alive.
The monophyly of the cohort Glires, has been disputed for many years. In the present, new genetic studies confirm the monophyly between the two orders, rodents and lagomorphs. Glires however, includes many ancestral, primitive and basal forms that cannot be stricly defined as a rodent or a lagomorph. Glires can be actually better divided to duplicidentata and simplicidentata. Duplicidentates (two tooth) includes the lagomorphs and other closely related forms (with two pair of front incisors) that are not part of the crown group lagomorpha. The same applies for simplicidentata (one tooth), Rodents and their closely allied forms. Inside simplicidentata, another order sister to Rodentia was established to lump some non-rodent simplicidentates called Mixodonta.
One monotypic genus of one of these obscure basal gliroids is Rhombomylus turpanensis. It is known from a lot of different specimens, also from different stages of ontogenic development. Its anatomy is derived to a certain extend, more so, than that of its very close relative Matutinia nitidilus, which possesses more primitive dentition and skull, and it is also smaller in size.
Here is my musculoskeletal reconstruction of the anatomy of Rhombomylus turpanensis. The postrcranial anatomy of this species remains largely obscure. It was likely an unspecialized terrestrial, non-cursorial form with some capabilities for jumping. The hindlimbs are much longer than the forelimbs.

And the life reconstruction:

Here is my another reconstruction of a close relative of Rhombomylus of a different genus, Matutinia nitidilus. I see these animals resembling perhaps the living pikas (Ochotonidae) or also the true rats (Rattus). Terrestrial herbivores, living in burrows and dens. They would also supply their diet with animal protein if given the change. These are my guesses of what would be their best modern analogue, althought their trophic ecology did not had to be exactly identical to the before-mentioned examples.

Basal gliroids and their closely related clades remain largely obscure. I hope more research will continue on this group to shed some light on these creatures.
References:
- SUYIN TING, JIN MENG, MALCOLM C. McKENNA, and CHUANKUEI LI “The Osteology of Matutinia(Simplicidentata, Mammalia) and Its Relationship to Rhombomylus,” American Museum Novitates2002(3371), 1-33, (21 June 2002). https://doi.org/10.1206/0003-0082(2002)371<0001:TOOMSM>2.0.CO;2
- JIN MENG, YAOMING HU, and CHUANKUI LI “THE OSTEOLOGY OF RHOMBOMYLUS (MAMMALIA, GLIRES): IMPLICATIONS FOR PHYLOGENY AND EVOLUTION OF GLIRES,” Bulletin of the American Museum of Natural History 2003(275), 1-247, (28 February 2003). https://doi.org/10.1206/0003-0090(2003)275<0001:TOORMG>2.0.CO;2
Tsaganomys altaicus
I present another short post and reconstruction of another not very well-known prehistoric tooth-digging rodent, of Early Oligocene.
So, here we have it, Tsaganomys altaicus. It belonged to the family Tsaganomyidae, comprising 3 genera and 5 species. Tsaganomys was native to Oligocene and possible early Miocene of Asia. It possesed a protrogomorphous zygomasseteric system (the most primitive type found in rodents) of the skull and a hystricomorphous mandible. It had massively developed almost circular zygomatic arches, large temporal fossa, prominent sagittal and occipital crests, hypsodont (high crowned) dentition and of course, procumbent incisors, among other traits.
The phylogenetic position and ancestry of tsaganomyids appears to be unresolved. Due to several superficial similarities, tsaganomyids were associated with bathyergids, cylindrodonts and hystricognaths. Now it’s clear that this group is not closely related to any of the before mentioned clades. For example, the bones of the middle ear, malleus and incus are not fused as in Bathyergidae, or for example the incisor microstructure is quite different from that of the cylindrodonts. Also, they lack other derived features common for hystricognaths. For now, tsaganomyids shall remain Rodentia incertae sedis.
For more detailed description, I highly suggest reading the original paper I referenced (under the drawing).
Here is my reconstruction of Tsaganomys altaicus. Missing postcranial proportions are based off of Bathyergus and soft tissues are based off of several different species of zokors (Myospalacinae).

References:
On Tsaganomyidae (Rodentia, Mammalia) of Asia (Banyue Wang, 2001) https://doi.org/10.1206/0003-0082(2001)317%3C0001:OTRMOA%3E2.0.CO;2
Rhizospalax poirrieri: a fossorial castorimorph of Europe
Tooth-digging has convergently evolved multiple times among unrelated clades of fossorial rodents throughout the history of the Cenozoic. Contrary to the mole-like digging in which the forelimbs act as shovels, tooth and head digging forms possess unspecialized, relatively small feet, most of the digging is done by the one pair of incisors and the head. One clade of these animals is the Spalacinae.
Based on almost identical shape of the mandible, Rhizospalax was once thought to be a spalacid, but detailed examination of its skull and dentition showed that it was of the sciuromorphous, castoroid type. It possessed a large P4, which when present in muroids, is definitely smaller than M1 (spalacids are muroids). The dentition of Rhizospalax was therefore 1.0.1.2/1.0.1.2 (last premolar substituted the first molar – P4, M1, M2) unlike a typical muroid which has only three true molars. Moreover, its molar pattern is less complex as in muroidea, indicating a different ancestry. Another trait that links this taxon to castorids is the shape and position of the infraorbital canal.
Fossorial beavers are known from Oligocene of North America. As a french find, Rhizospalax is a proof that, tooth-digging castorimorphs likely once inhabited the whole holarctic realm, and occupied similar ecological niches now filled by the still-living Bathyergidae, arvicoline tribe Ellobuisini and Spalacidae.
Here is shown my life reconstruction of Rhizospalax poirrieri. Missing components are based due to ecomorphological similarities, on Spalax, Tachyoryctes and Capacikala.

Literature:
- A new rodent from the Upper Oligocene of France. Bulletin of the AMNH; v. 41, article 18. Gerrit Smith Miller, James Williams Gidley, B Poirrier, Claude Gaillard
Cave bears of Europe
Succeeding the Eemian or the Last Interglacial, the Last Glacial period (115, 000 -11, 000 years ago) began. It is a well known fact, that during this time, cave bears (Ursus ex. gr spelaeus) were widespread in Eurasia. This lineage was closely related to recent brown and polar bears. Althought rivaling their extant relatives in terms of dimensions (and on average being heavier), cave bears were specialized herbivores. It can be deduced from numerous factors (isotope analysis, tooth microwear…). There is no doubt they would be a formidable adversary when confronted with our ancestors.

Cave bears (Ursus ex. gr spelaeus) were very diverse, with different forms living on different localities and having different prefences (altitude and perhaps also plant types..). In central and eastern Europe, Ursus ingressus was one of the largest forms, occupying habitats below the ranges of the smaller, high alpine subspecies such as Ursus spelaeus ladinicus and Ursus spelaeus eremus.
As it has been found, U. ingressus and U. s. eremus were living sympatrically for 15,000 years in Austria, with no apparent interbreeding. This is an evidence, that they must have niche partitioned and were quite different ecologically.
There were also a lot of different forms. For example, the classical Ursus spelaeus spelaeus was found in western Europe.
The brown bears (Ursus arctos) lived alongside cave bears, but were more carnivorous than they are today. This was to avoid competition.
Literature used:
- Explanations from Prof. Martin Sabol, PhD.